Sign of Relief for 1.33M Kenyans With HIV as Twice-a-Year Lenacapavir Rollouts Offer New Prevention Options
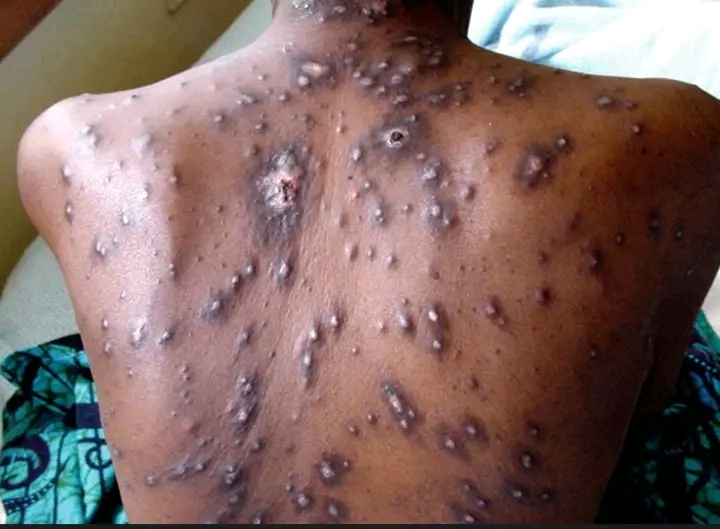
For the 1.33 million Kenyans living with HIV, prevention has long been a daily struggle with oral medication, adherence challenges, and social stigma. The introduction of Lenacapavir, a twice-yearly injectable drug, offers a promising alternative that could simplify prevention for high-risk populations.
The United States, in partnership with Gilead Sciences and the Global Fund to Fight AIDS, Tuberculosis, and Malaria, has begun rolling out initial doses to countries in southern Africa, with Kenya included in upcoming distribution plans. This initiative represents a critical development in the fight against HIV in a country where new infections continue to challenge health systems and public health interventions Stats Kenya, 2025.
Globally, HIV remains a persistent public health concern despite decades of progress in prevention and treatment, with an estimated 1.3 million new infections reported in 2024. Long-acting preventive therapies like Lenacapavir are designed to address adherence issues that have limited the effectiveness of daily oral PrEP, particularly among women, adolescents, and pregnant or breastfeeding mothers.
The global rollout strategy leverages partnerships with international organizations and pharmaceutical companies to ensure that the drug reaches populations most in need in a timely manner. Expanding access to innovative interventions is particularly urgent in sub-Saharan Africa, where HIV prevalence and incidence remain high and progress in reducing new infections has plateaued in several countries.
M-Pesa’s Role in Kenya’s Fintech Growth
In Kenya, approximately 1.33 million people are living with HIV, representing a prevalence rate of 3.03 percent according to the latest national statistics. Of this population, women account for the majority, with 867,571 individuals living with the virus, reflecting the disproportionate impact on female populations across the country.
New infections remain a critical concern, with 20,105 cases recorded in 2025, underscoring the need for more effective prevention strategies and broader access to interventions like Lenacapavir. The introduction of a twice-yearly injectable provides a significant opportunity to reduce the burden of daily adherence while potentially increasing uptake among populations that face social or logistical barriers to existing prevention methods.
For many Kenyans, particularly young mothers and adolescents, receiving a six-monthly injection rather than taking daily pills can remove both practical and social barriers to prevention. Health workers have emphasized that reducing the frequency of medication intake may improve adherence rates, lessen stigma, and enhance the effectiveness of national prevention programs.
Pilot programs in Eswatini and Zambia are already providing critical insights into how populations respond to long-acting HIV prevention strategies, which will inform Kenya’s planned rollout. These early lessons will help determine how best to integrate Lenacapavir into routine healthcare delivery, particularly in areas with limited infrastructure.
The clinical profile of Lenacapavir demonstrates its potential to improve prevention outcomes by offering durable protection through two annual injections. Compared to daily oral PrEP, this approach mitigates the risk of missed doses, which has historically contributed to continued transmission and treatment failures.
The drug’s mechanism, which allows it to remain effective in the body for six months, represents a significant advancement in HIV biomedical prevention technology. By lowering the frequency of administration, Lenacapavir could also reduce the psychological and social burden associated with daily medication, particularly in communities where HIV stigma remains high.
The U.S.-backed initiative to deliver Lenacapavir to African countries, including Kenya, is part of a broader strategy to strengthen HIV prevention in low- and middle-income nations. Approximately 1,000 doses have already been delivered to Eswatini and Zambia, with plans to scale up to two million doses in the coming years.
This strategy emphasizes targeted distribution to high-burden populations, including women, adolescents, and pregnant or breastfeeding individuals, ensuring that interventions reach those most at risk. By coordinating with international partners, Kenya can leverage these resources to expand its prevention efforts while maintaining the quality and safety of the rollout.
Experts caution, however, that innovation alone is insufficient to achieve significant reductions in HIV transmission. Sustainable impact will require investments in health systems, including clinic staffing, cold-chain logistics, and robust monitoring and evaluation frameworks.
Policy frameworks and community engagement strategies must also ensure equitable access, particularly for marginalized populations who have historically been underserved by prevention programs. Without addressing these structural and social determinants, the benefits of Lenacapavir could remain concentrated in urban or well-served areas, leaving rural and high-risk communities behind.
Lenacapavir is a long-acting antiretroviral medication designed to prevent HIV infection by targeting the virus’s capsid, a protein shell essential for viral replication. Unlike daily oral PrEP, it is administered via subcutaneous or intramuscular injection only twice a year, which significantly reduces adherence challenges and provides sustained protection.
Clinical trials have shown that the drug is generally well tolerated, with mild injection site reactions being the most common side effect, while systemic side effects such as headache or nausea remain rare. In Kenya, the rollout is expected to be provided at no cost to eligible high-risk populations through public health programs supported by the Global Fund and government partnerships.
The mechanism of Lenacapavir is distinct from most oral prevention drugs that target reverse transcriptase, as it inhibits the viral capsid, preventing HIV from establishing infection in the body. This difference makes it a particularly valuable addition to existing prevention strategies, as it can be effective in individuals who may struggle with daily pill adherence or who have developed resistance to other preventive medications.
Healthcare providers emphasize that Lenacapavir should be administered in clinical settings under trained supervision to monitor for any potential reactions, with follow-up visits scheduled to maintain the six-month dosing schedule. By combining long-lasting pharmacological effects with user-friendly administration, the drug aims to enhance adherence and reduce new infections among populations most at risk.
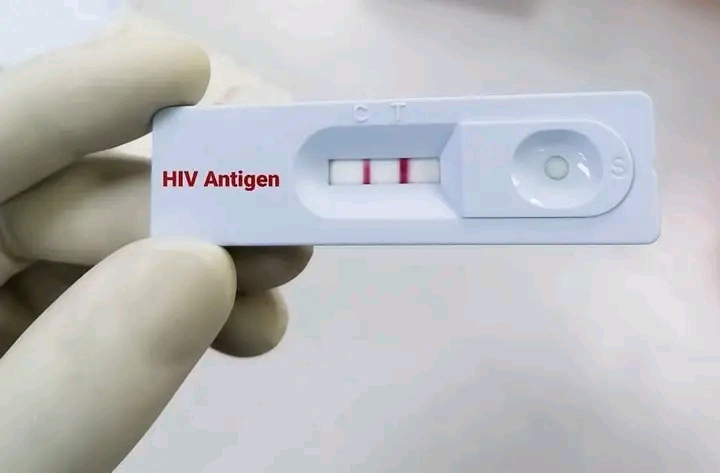
For people living with HIV or at high risk, understanding the practical details of Lenacapavir is critical. The injections are designed to be given by trained nurses or clinicians in public health facilities, with clear guidelines on dosage and timing to maintain protective drug levels in the body.
While it is not a cure and does not eliminate HIV transmission risk entirely, Lenacapavir can significantly reduce the likelihood of infection when used as part of comprehensive prevention strategies including condom use, regular testing, and adherence support. Patients are advised to report any side effects promptly, although current trial data indicate the drug is safe, effective, and well tolerated, making it a promising option in Kenya’s ongoing efforts to expand access to HIV prevention.
“Lenacapavir offers a twice-yearly option that reduces adherence challenges and enhances protection for high-risk populations.”
This article was prepared by the Ramsey Focus Analysis Desk, based on verified reports, independent analysis, and insights to ensure balanced coverage.


















2 Responses
Very nice information
Keep it up
Thanks